
The first atomic radius periodic trend is that atomic radii increase as you move downwards in a group. What are the periodic trends of atomic radius? What is the atomic radii of fluorine and neon?Ītomic radii of florine and neon The atomic radii of fluorine and neon in the Angstrom unit are 0.75 and 1.60 respectively. The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Which of the following order of radii is correct? While going down the group the atomic radii goes on increasing, this is because new shells are added to the atoms of the elements as we go down from top to bottom in a group. What is the periodic trends observed in the variation of atomic radii down a group? Hence the correct order is O2−>F−>Na+>Mg2+. What is the correct order of ionic radius? Which one of the following is the correct increasing order of the magnitude of ionic radii CE³+ LA³+ PM³+ and YB³+? The atomic radius increases when we go down a group because of the addition of an extra shell. In general, the atomic radius decreases as we move from left to right in a period with an increase in the nuclear charge of the element. One atomic radius trend occurs as you move left to right across the periodic table (moving within a period), and the other trend occurs when you move from the top of the periodic table down (moving within a group). The correct increasing order of ionic radii of the following Ce3+, La3+, Pm3+ and Yb3+ is. Which of the following is the correct order of radius of La3 Ce3 Pm3 Yb3 +? Across a period, effective nuclear charge increases as electron shielding remains constant. In general, atomic radius decreases across a period and increases down a group. What is the trend for atomic radius radii? 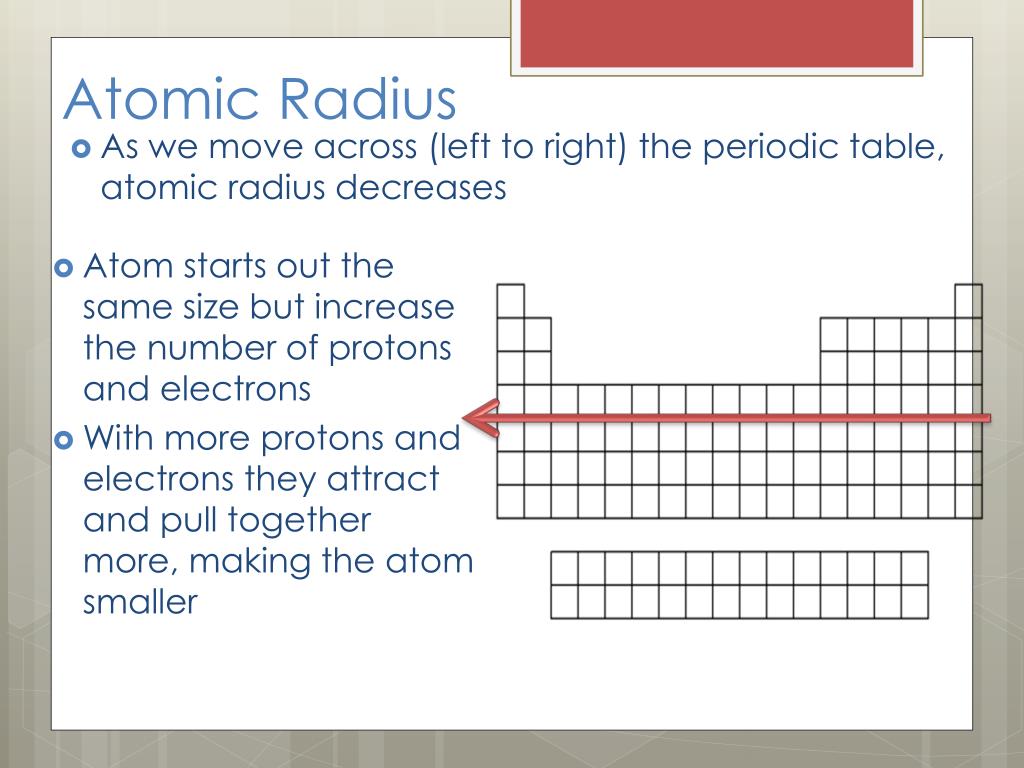
How do atomic radii change across a period?.What are the periodic trends of atomic radius?.What is the atomic radii of fluorine and neon?.What is the periodic trends observed in the variation of atomic radii down a group?.What is the correct order of ionic radius?.Which one of the following is the correct increasing order of the magnitude of ionic radii CE³+ LA³+ PM³+ and YB³+?.What are the 2 trends in atomic radius?.Which of the following is the correct order of radius of La3 Ce3 Pm3 Yb3 +?.What is the trend for atomic radius radii?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
